Introduction
Human mesenchymal stromal cells (hMSCs) are considered the “workhorse” of Regenerative Medicine (RegenMed). hMSCs are a critical starting material in a growing variety of established and emerging RegenMed products, including cellular therapies, cell-based gene therapies, hMSC-derived extracellular vesicles (EVs), and bioprinted engineered tissues (Olsen, 2018). Accordingly, there have been greater than 100 clinical trials initiated each year since 2011 using hMSC from various sources across a host of indications and therapeutic strategies (clinical trials.gov). hMSCs have benefitted from having an excellent safety profile, and there have been nine products approved globally over the last 10 years. This growth in the use of hMSCs in a variety of product types has created the opportunity to standardize the supply chain and provide economies of scale for a rapidly growing industry. RoosterBio was founded to industrialize and standardize the RegenMed supply chain and to radically simplify the incorporation of living cells into therapeutic product development. Our goal is to have the same impact on the RegenMed industry that Intel had on the computer industry.
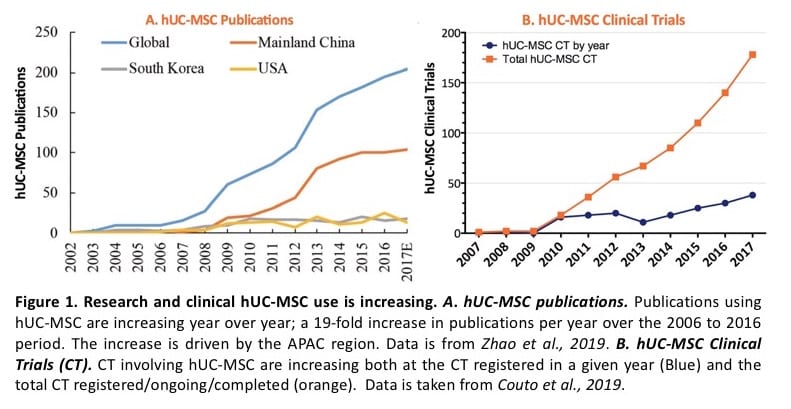
The use of human Umbilical Cord-derived MSCs (hUC-MSCs) in research and clinical trials has grown rapidly over the last 10 to 15 years with the quickest adoption in APAC (Figure 1, Davies, 2017; Zhao, 2018; Moll, 2019). hUC-MSC publications per year increased 19-fold from 2006 to 2016 (Zhao, 2018). Clinical trials with hUC-MSCs have shown a similar growth to publications. hUC-MSCs are the second most used hMSC type in clinical trials (Moll, 2019), and 178 clinical trials using hUC-MSCs were registered, are ongoing, or were completed between 2007 and 2017 (Couto, 2019). In fact, >30% of hMSC trials registered in 2019 used hUC-MSCs as the cell source. These drive the need for hUC-MSCs to use in product development. Until now, IP surrounding hUC-MSCs has been a primary roadblock to the widespread adoption of hUC-MSCs. We have collaborated with leaders in Wharton’s Jelly/umbilical cord hMSCs at Tissue RegenerationTherapeutics Inc. (TRT) and have brought to market a complete bioprocess cell and media system. RoosterBio’s hUC-MSCs are available for licensing and provided in scalable formulations with cGMP-compatible processes that enable anyone to obtain hUC-MSCs in the numbers needed for incorporation into RegenMed product development.
Until now RoosterBio has paired our batch (2D) and fed-batch (3D bioreactor) bioprocess media systems with hMSCs from two sources: adipose-derived (hAD-MSC) and bone marrow-derived (hBM-MSC and Xeno-Free hBM-MSC). RoosterBio’s launch of our XF hUC-MSCs (RoosterVial™-hUC-10M-XF and RoosterKit™-hUC-1M-XF) introduced the first umbilical cord-derived hMSC in the market designed to meet the quality and volume needs of today’s translationally focused cell therapy product developers. See RoosterBio’s hMSC product lines here.
RoosterBio’s RoosterVial-hUC-MSC-XF and RoosterNourish™-MSC-XF cell and medium bioprocess system has several key advantages over the limited number of suppliers of perinatal hMSCs. Being XF, and manufactured with RoosterBio’s existing cGMP-compatible processes, our system is the only hUC-MSC commercially available with a clear line of sight to clinical translation. Additionally, other suppliers (a) provide low cell number vials at a high price per M cells, (b) supply serum-based cells, or (c) require specialized, non-scalable culture vessels. Finally, RoosterBio provides first-in-class characterization of hMSC key quality attributes (PDL, identity, expansion potential) and functional assays (cytokine secretion, trilineage differentiation, immunomodulation).
Results
 For standardized, high-quality hUC-MSCs, we put in place stringent design requirements, accounting for expected donor-to-donor variability, hUC-MSCs’ known high expansion rates, and the ISCT criteria for hMSCs (Carmen, 2012; Dominici, 2006; Krampera, 2013; Bravery, 2013, Davies, 2017).
For standardized, high-quality hUC-MSCs, we put in place stringent design requirements, accounting for expected donor-to-donor variability, hUC-MSCs’ known high expansion rates, and the ISCT criteria for hMSCs (Carmen, 2012; Dominici, 2006; Krampera, 2013; Bravery, 2013, Davies, 2017).
As shown below, RoosterBio’s hUC-MSCs meet these stringent criteria. Our hUC-MSCs, RoosterVial-hUC:
- Have very high expansion rates in a 2D batch culture
- Meet ISCT criteria for MSC: (a) cell identity, (b) immunomodulation, and (c) trilineage differentiation
- Are characterized for functional potency (angiogenic cytokine secretion)
- And have unprecedented, quantified productivity metrics (Carmen, 2012; Dominici, 2006; Krampera, 2013; Bravery, 2013)
Unlike other hMSC vendors, we perform our quality assays at the PDL (i.e. level of expansion) that we recommend our customers use our cells. So, all assays presented here were performed with RoosterBio hUC-MSCs that have been expanded for 2 passages (~10 PDL) from product vials (RoosterVial-hUC-10M-XF, C43002 or RoosterVial-hUC-10M-XF, C43002) in RoosterNourish-MSC-XF medium, following RoosterBio’s process recommendations.
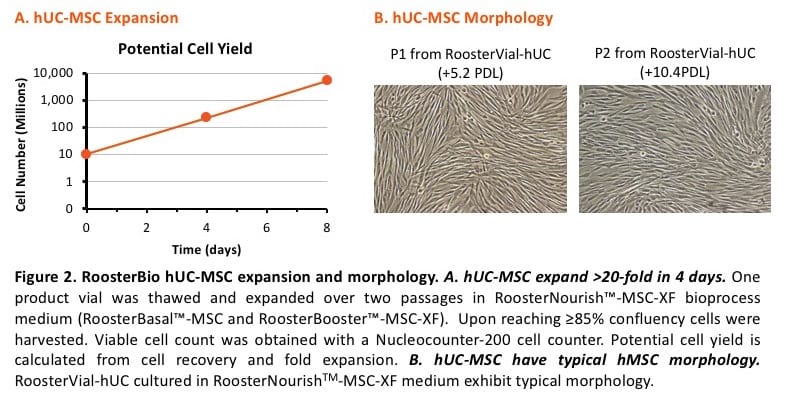
Cell expansion. A key characteristic of RoosterBio hMSC bioprocess systems is rapid cell expansion. For example, RoosterBio’s hBM-MSC-XF in RoosterNourish™-MSC-XF are guaranteed for >10-fold expansion within 5-7 days. See our popular blog post on PDLs. Our hUC-MSCs exceed this growth rate with a typical expansion of >20-fold in 4 days (see Figure 2A for a representative RoosterBio product donor). We see variability across donors, but donors are typically harvested at greater than 80,000 cells/cm2, after plating at ~3,000 cells/cm2, within 4 days (Figure 2B). So, a vial of 10M hUC-MSC (RoosterVial-hUC-10M-XF) grown over 2 passages has a potential yield of greater than 5B hUC-MSC in 8 to 10 days, leading to significant economic benefits (described below).
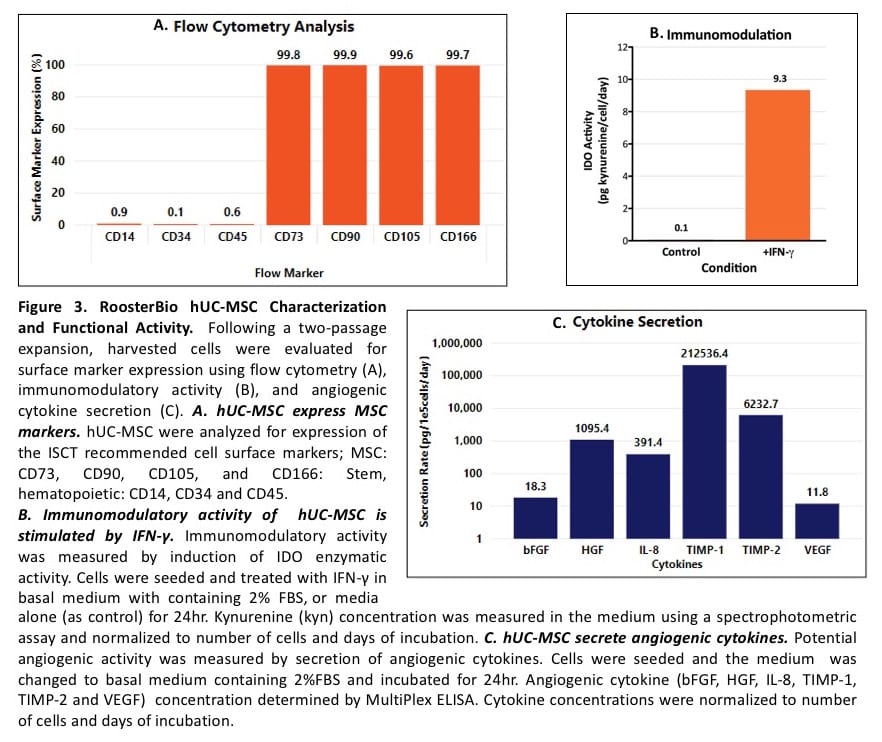
Cell surface marker expression. Flow cytometry analysis for the ISCT-recommended cell surface markers was done for RoosterVial-hUC in RoosterNourish-MSC-XF following a 2 passage expansion. The hUC-MSC population displayed the typical hMSC surface marker expression — they were low (<5% positive) for the stem and hematopoietic cell markers, CD14, CD34, and CD45, and >90% positive for hMSC markers CD73, CD90, CD105, and CD166 (Figure 3A).
Immunomodulatory function. Immunomodulation is an essential part of the in vivo therapeutic role(s) of hMSC (Krampera 2013). The immunomodulatory potential of hMSC is assayed by activation of indoleamine 2,3-dioxygenase (IDO) activity by the pro-inflammatory cytokine IFN-γ and measuring kynurenine (kyn), an immunosuppressive product of the IDO reaction, in the cell culture supernatant. hUC-MSC were expanded in RoosterNourish-MSC-XF for 2 passages, harvested, plated, and treated with IFN-γ. The kynurenine concentration in the cell supernatant was measured using a spectrophotometric assay. Like other hMSC types, hUC-MSC showed low basal IDO activity that was inducible by IFN-γ treatment (Figure 3B). We see variability in induced IDO activity across UC donors (not shown) which will be a subject of a subsequent blog. Importantly, these data show that RoosterBio’s hUC-MSCs meet the ISCT criterion by demonstrating immunomodulatory activity.
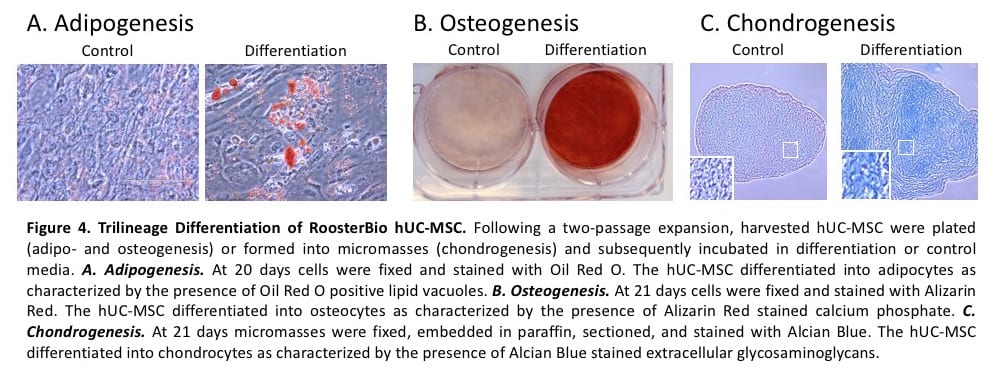
Trilineage differentiation. A hallmark characteristic of hMSCs and ISCT criterion is differentiation in vitro to adipocytes, osteocytes, and chondrocytes (Dominici, 2006; Krampera, 2013). RoosterBio’s hUC-MSCs expanded 2 passages from the working cell bank in RoosterNourish-MSC-XF were harvested, plated, and incubated in Adipogenesis, Osteogenesis, or Chondrogenesis Media. After 18-21 days in culture, staining for adipogenic, osteogenic, and chondrogenic differentiation potential was performed. As shown in Figure 4, RoosterBio’s hUC-MSC differentiated to fat, bone, and cartilage.
Potency: Angiogenic cytokine secretion. hMSCs achieve their therapeutic effects by secreting a plethora of biomolecules that influence many biologic processes (Murphy 2013). As a measure of hUC-MSC potency, we assayed the secretion of the angiogenic cytokines bFGF, HGF, TIMP 1, TIMP 2, IL-8, and VEGF via a multiplexed ELISA analysis (Figure 3C). When multiple UC donors are screened we expect hUC-MSCs to differ in their cytokine secretion profile from hAD-MSC and hBM-MSC (some of the cytokines may be higher or lower, etc).
Potency: Extracellular vesicle (EV) Production. Due to their similar therapeutic effects to MSCs and potential as a key bioactive agent in regenerative medicine applications, MSC-derived EVs are being increasingly investigated in pre-clinical research and as a clinical therapy for a broad range of indications (Elahi, 2019). However, EV production for these studies is limited by functional cell number. Our bioprocess media systems for cell growth (RoosterNourish-MSC) and EV production (RoosterCollect™-EV) are an efficient, scalable system to produce EVs from hMSC. We have preliminary studies showing robust EV production from our hUCs-MSC (the subject of a future blog).
hMSC Tissue origin differences. As expected, we see variability in expansion, immunomodulatory activity, differentiation, and angiogenic cytokine secretion across UC donors (not shown). hUC-MSCs also have tissue-to-tissue differences when compared to other MSC types. Thus, RoosterBio provides at least 3 donors for each of our hMSC tissue sources allowing customers to screen for the optimal tissue type and donor for their specific application or target indication. The relative tissue-specific strengths of hMSC types as well as emerging application areas will be discussed in subsequent blogs.
Productivity Metrics. Due to the very high expansion rates discussed above, the RoosterVial-hUC, RoosterNourish-MSC-XF bioprocess system has unprecedented quantifiable productivity metrics. The expansion productivity in 2D (e.g. multi-layer vessels) is ~800 M cells/L of media, an increase over the already high productivity of our hBM-MSC-XF (200-400 M cells/L). With our hUC-MSC cell and media system at least 5B cells can be produced in 8-10 days for an investment of ~$6 to $8K. This productivity increase is also demonstrated in 3D bioreactor and exosome generation (and will be highlighted in future blog posts).
Conclusion
RegenMed will source hMSC from multiple tissue types because each hMSC type will show origin-specific strengths; such as variations in expansion capability, immunomodulatory potential, angiogenic cytokine secretion, tissue-specific differentiation, and EV production and characteristics. Therefore, we are introducing XF hUC-MSC (RoosterVial-hUC-XF) to expand our portfolio. Having hMSC from multiple tissue sources supports our customer screening of donor AND tissue source to find their best hMSC. Paired with RoosterBio’s bioprocess media system, RoosterNourish-MSC-XF, RoosterVial-hUC reaches new heights in hMSC 2D batch and 3D bioreactor fed-batch expansion productivity in terms of both M cells/L media and overall cost, while generating larger volumes (Billions) of hMSCs and EVs. Additionally, RoosterBio provides first-in-class characterization of hUC-MSC key quality attributes (PDL, identity, expansion potential) and functional potential (cytokine secretion, differentiation, immunomodulation). Finally, RoosterBio’s scalable, cGMP-compatible, and ready-to-implement process recommendations simplify and accelerate the path through hUC-MSC-based product development to clinical implementation.
References
Bravery CA, Carmen J, Fong T, Oprea W, Hoogendoorn KH, Woda J, Burger SR, Rowley JA, Bonyhadi ML, & Van’t Hof W (2013) Potency assay development for cellular therapy products: an ISCT review of the requirements and experiences in the industry. Cytotherapy 15(1):9-19. http://www.ncbi.nlm.nih.gov/pubmed/23260082
Carmen J, Burger SR, McCaman M, & Rowley JA (2012) Developing assays to address identity, potency, purity, and safety: cell characterization in cell therapy process development. Regenerative Medicine 7(1):85-100. http://www.ncbi.nlm.nih.gov/pubmed/22168500
Couto PS, Shatirishvili G, Bersenev A, Verter F. (2019) First decade of clinical trials and published studies with mesenchymal stromal cells from umbilical cord tissue. Regenerative Medicine 14(4):309-319. http://www.ncbi.nlm.nih.gov/pubmed/31070115
Davies JE, Walker JT, Keating A. (2017) Concise Review: Wharton’s Jelly: The Rich, but Enigmatic, Source of Mesenchymal Stromal Cells. Stem Cells Transl Med. 6(7):1620-1630. https://www.ncbi.nlm.nih.gov/pubmed/28488282
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop D, & Horwitz E (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8(4):315-317. http://www.ncbi.nlm.nih.gov/pubmed/16923606
Elahi FM, Farwell DG, Nolta JA, Anderson JD (2019) Concise Review: Preclinical Translation of Exosomes Derived from Mesenchymal Stem/Stromal Cells. Stem Cells. Epub ahead of print. https://www.ncbi.nlm.nih.gov/pubmed/31381842
Krampera M, Galipeau J, Shi Y, Tarte K, Sensebe L, & MSC Committee of the International Society for Cellular Therapy (ISCT) (2013) Immunological characterization of multipotent mesenchymal stromal cells–The International Society for Cellular Therapy (ISCT) working proposal. Cytotherapy 15(9):1054-1061. http://www.ncbi.nlm.nih.gov/pubmed/23602578
Moll G, Ankrum JA, Kamhieh-Milz J, Bieback K, Ringdén O, Volk HD, Geissler S, Reinke P (2019) Intravascular Mesenchymal Stromal/Stem Cell Therapy Product Diversification: Time for New Clinical Guidelines. Trends Mol Med. 25(2):149-163. https://www.ncbi.nlm.nih.gov/pubmed/30711482
Murphy MB, Moncivais K, & Caplan AI (2013) Mesenchymal stem cells: environmentally responsive therapeutics for regenerative medicine. Experimental & Molecular Medicine 45:e54. http://www.ncbi.nlm.nih.gov/pubmed/24232253
Olsen TR, Ng KS, Lock LT, Ahsan T, Rowley JA (2018) Peak MSC – are we there yet? Front. Med., 21 June 2018. https://www.ncbi.nlm.nih.gov/pubmed/29977893
Zhao J, Yu G, Cai M, Lei X, Yang Y, Wang Q, Zhai X (2018) Bibliometric analysis of global scientific activity on umbilical cord mesenchymal stem cells: a swiftly expanding and shifting focus. Stem Cell Research & Therapy 9(32). https://www.ncbi.nlm.nih.gov/pubmed/29415771
