MSC-EVs Emerge as a Cell-Free Therapy
Extracellular vesicle (EV) interest continues to increase as more evidence emerges about the ability of these lipid-bilayer membrane vesicles to elicit specific responses from recipient cells. EVs are secreted by most known cell types, including MSCs. Recently, many effects of MSC-based therapeutics have been attributed to their paracrine factors which includes MSC-derived EVs (1, 2). In particular, MSC-derived EVs have been shown to recapitulate therapeutic effects of MSCs in graft-versus-host disease (3)and myocardial ischemia (4), among others. Moreover, EVs derived from MSCs benefit from MSCs’ well-defined safety profile, with MSCs having been used in over 900 clinical trials. Given their therapeutic potential, EVs are on the rise as a novel clinical therapy for a broad range of applications. This interest is reflected in the high number of peer-reviewed publications in the past 10 years mentioning EVs (over 15,000), with 700 specifically on MSC-EVs (PubMed Search Results Oct 2019) and the larger presence of EVs at cell therapy conferences.
MSC-EVs as Drug Delivery Vehicles
In addition to their use as a cell-free therapy, there is also significant interest in using EVs as drug delivery vehicles. EVs are natural carriers of bioactive cargo such as proteins and RNA, which are protected by the lipid-bilayer membrane. Research efforts have focused on both exogenous loading of biological cargo and manipulating parent cells to engineer vesicles that contain cargo of interest.
RoosterBio EVs
RoosterBio MSC-EVs were evaluated based on guidance for defining EVs, as published by the International Society for Extracellular Vesicles (5). Particles collected from the conditioned medium of RoosterBio MSCs are in the EV size range, contain expected proteins and small RNAs, and have bioactivity in a wound-healing assay (Figure 1).
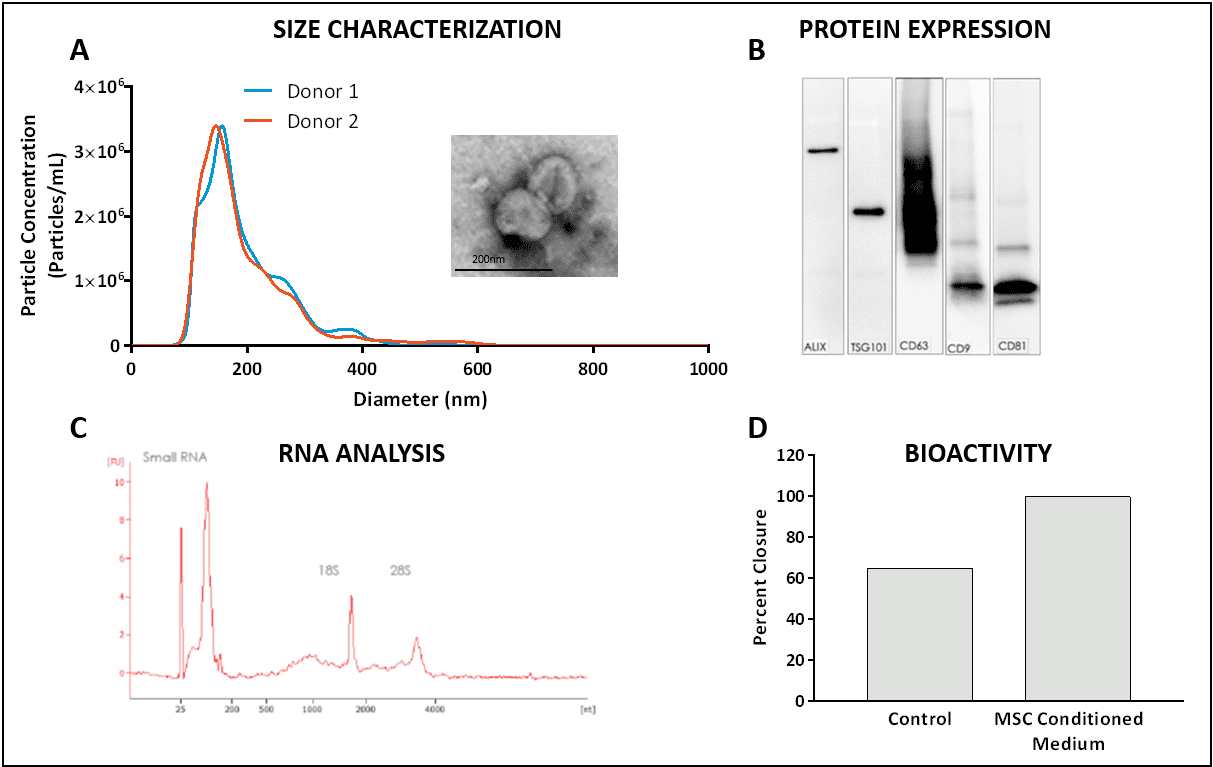
Figure 1A. Particles collected from RoosterBio MSCs have diameters in the size range of 50 to 250 nm as measured by Nanosight and TEM. B. Western Blot shows EVs express expected proteins: ALIX, TSG101, CD63, CD9, and CD81. C. Collected EVs contain primarily small RNA. D. Conditioned medium has bioactivity in a standard in vitro wound healing scratch test.
Challenges Facing the Field
While EVs hold much promise as a cell-free therapy or drug delivery vehicle, there are some key challenges in the translation of successful EV therapies, including generating the needed number of EVs. In our next blog we will discuss some of these key challenges that need to be addressed to enable the success of EV therapies and RoosterBio’s progress in meeting the needs for EV product development, clinical trials, and commercial therapies.
References
- Phinney DG, Pittenger MF. (2017) Concise Review: MSC-Derived Exosomes for Cell-Free Therapy. Stem cells 35(4):851-858. PubMed
- Caplan AI, Correa D. (2011) The MSC: an injury drugstore. Cell stem cell 9(1):11-15. PubMed
- Kordelas L, Rebmann V, Ludwig AK, Radtke S, Ruesing J, Doeppner TR, et al. (2014) MSC-derived exosomes: a novel tool to treat therapy-refractory graft-versus-host disease. Leukemia 28(4):970-973. PubMed
- Lai RC, Arslan F, Lee MM, Sze NS, Choo A, Chen TS, et al. (2010) Exosomes secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem cell research 4(3):214-222. PubMed
- Lötvall J, Hill AF, Hochberg F, Buzás EI, Di Vizio D, Gardiner C, et al. (2014) Minimal experimental requirements for definition of extracellular vesicles and their functions: a position statement from the International Society for Extracellular Vesicles. J Extracell Vesicles 3:26913. PubMed
