prcRoosterReplenish™-MSC-CC
A cGMP fed-batch bioreactor feed with pathogen reduced components for hMSC manufacturing.
Engineered to enable scalable, 3D bioreactor hMSC GMP manufacturing.
Recommended Products
Overview
prcRoosterReplenishTM-MSC-CC is a xeno-free (XF), GMP concentrated bioreactor feed for use in fed-batch hMSC manufacturing bioreactor processes to eliminate media exchanges during rapid expansion of human MSCs for clinical allogeneic and autologous therapeutic cell manufacturing.
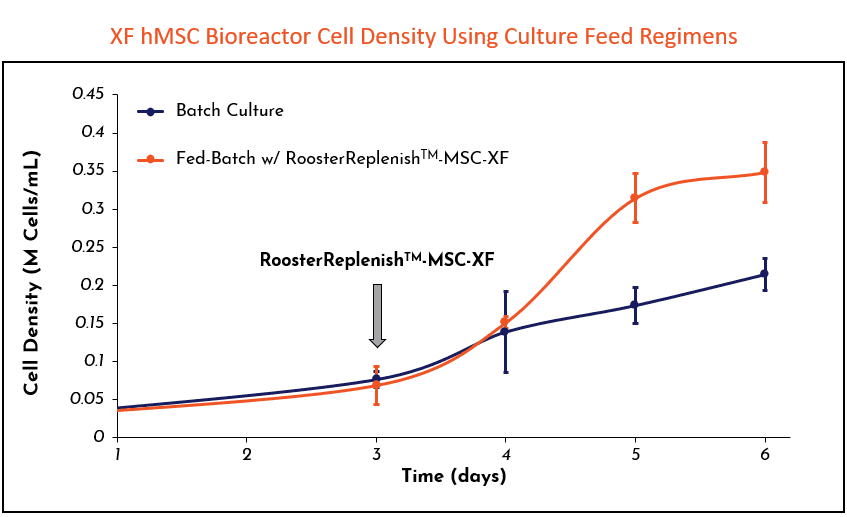
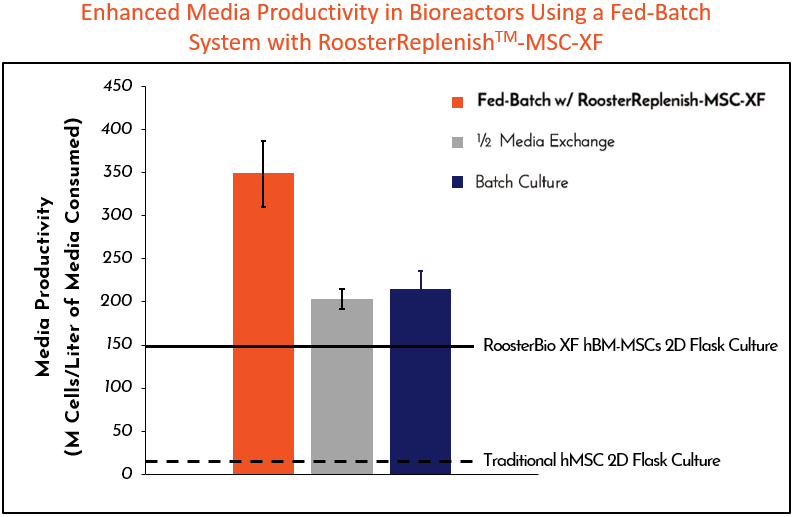
Engineered to achieve high cell growth in scalable (>50L) microcarrier-based suspension cultures when paired with our CC RoosterVial™-MSCs and prcRoosterNourish-MSC-CC while reducing contamination risks associated with in-process manipulation. prcRoosterReplenish-MSC-CC boosts media productivity (millions of cells produced per Liter of media consumed) by up to 70%, with each 100 ml bagged unit supplementing 5L of bioreactor expansion media.
CliniControl™ bioprocess media are manufactured with methods and controls that conform with current Good Manufacturing Practices (cGMP) and are supported for use as an ancillary material by a Type II Master File on file with the FDA. Time spent on investigational new drug (IND) submissions is significantly reduced by facilitating CMC documentation.
Product Features
- Xeno-free | cGMP | Pathogen Reduced (prc)*
- Enables a fed-batch process for scalable (>50L) 3D bioreactor hMSC culture.
- Each 100 ml bagged unit supplements 5L of prcRoosterNourish-MSC-CC in bioreactor setting.
- Eliminate media exchange by addition on Day 3 of bioreactor culture.
- Streamlines culture processes & reduces processing time and labor.
- Maintains stable nutrient and waste bio-profiles through 7 days of culture; comparable to batch culture.
- Mitigates risk of contamination by minimizing in-process culture manipulation.
- Available in custom high-volume bagged and non ‘prc’ formulations.
- Supported by US FDA Type II Master Files – available for cross reference
Intended Use: For Further Manufacturing Use Only. Not intended for diagnostic use or as an excipient for direct human or veterinary therapeutic use.
*’prc’ denoted products have been manufactured using pathogen reduced components to meet evolving regulatory needs and standards.
Protocols and Information
Login to search Certificates of Analysis and Quality Control Briefs.