 After the National Institute for Standards and Technology (NIST) identified upcoming challenges in the U.S. biopharmaceutical manufacturing landscape (see document here), the National Institute for Innovation in Manufacturing Biopharmaceuticals (NIIMBL) was formed to help solve these challenges through the formation of a public-private partnership between industry, government, academia, and non-profits. The goal of NIIMBL is to accelerate Biopharmaceutical manufacturing innovation, support the development of standards that enable more efficient and rapid manufacturing capabilities, and educate and train a world-leading Biopharmaceutical manufacturing workforce to maintain the United States’ global lead and competitiveness in this industry. NIIMBL will be leveraging a $70 million investment from NIST, with at least $129 million more in funds from private partners. One of the great aspects of this institute is that they are encouraging partnerships between large companies and smaller or medium-sized companies (called Small to Medium Enterprises “SME”), which will be sure to bolster innovation and streamline commercialization and implementation of new technologies. Kelvin Lee, the NIIMBL Institute Director, did a fabulous job taking the lead on organizing the Consortium’s first National Meeting, where members from the United States Congress, Directors from the Food and Drug Administration, and many executive-level industry representatives were invited to speak about the importance of manufacturing sciences and the current challenges we are facing as an industry. I had the opportunity to represent RoosterBio as an SME at this inaugural NIIMBL National Meeting, and I gave a talk in the “Rapid Fire” SME Innovation Showcase, as well as presented some of our work on how we are radically shortening the development timelines of Biopharmaceuticals that include a stem cell-derived component.
After the National Institute for Standards and Technology (NIST) identified upcoming challenges in the U.S. biopharmaceutical manufacturing landscape (see document here), the National Institute for Innovation in Manufacturing Biopharmaceuticals (NIIMBL) was formed to help solve these challenges through the formation of a public-private partnership between industry, government, academia, and non-profits. The goal of NIIMBL is to accelerate Biopharmaceutical manufacturing innovation, support the development of standards that enable more efficient and rapid manufacturing capabilities, and educate and train a world-leading Biopharmaceutical manufacturing workforce to maintain the United States’ global lead and competitiveness in this industry. NIIMBL will be leveraging a $70 million investment from NIST, with at least $129 million more in funds from private partners. One of the great aspects of this institute is that they are encouraging partnerships between large companies and smaller or medium-sized companies (called Small to Medium Enterprises “SME”), which will be sure to bolster innovation and streamline commercialization and implementation of new technologies. Kelvin Lee, the NIIMBL Institute Director, did a fabulous job taking the lead on organizing the Consortium’s first National Meeting, where members from the United States Congress, Directors from the Food and Drug Administration, and many executive-level industry representatives were invited to speak about the importance of manufacturing sciences and the current challenges we are facing as an industry. I had the opportunity to represent RoosterBio as an SME at this inaugural NIIMBL National Meeting, and I gave a talk in the “Rapid Fire” SME Innovation Showcase, as well as presented some of our work on how we are radically shortening the development timelines of Biopharmaceuticals that include a stem cell-derived component.
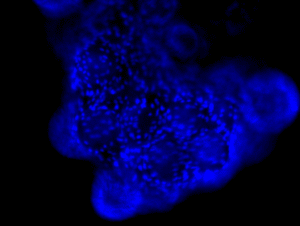 |
| Confluent hMSCs on Solohill microcarriers. |
Among the many relevant talks given throughout the day, one specifically caught my attention. In his talk titled “Key Process & Assay Challenges in Cell Therapy Development,” Greg Russotti (Vice President, Technical Operations at Celgene Cellular Therapeutics) laid out challenges in the upscaled manufacturing of human mesenchymal stem cells (hMSCs). To meet the pressing need for economical manufacturing of hMSCs at clinically- and commercially- relevant scales, researchers have turned to single-use bioreactor systems that have successfully been used to manufacture other biomolecules, such as monocolonal antibodies which make up the lion’s share of blockbuster pharmaceuticals. However, unlike small molecule or large molecule production, cell therapy products are living, breathing cells, which presents unique bioprocessing constraints and challenges. Greg noted that there are technologies based on monoclonal production that can expand hMSCs in large quantities, like using 3D microcarrier-based bioreactor systems, but there are still many manufacturing innovations required before these manufacturing platforms can support a commercial cell therapy product.
The technology gaps that he specifically mentioned for upscaled hMSC manufacturing were downstream processing technologies, specifically the unit operations related to:
a. detaching (or harvesting) the cells from microcarriers without damaging or changing the cells in any way,
b. separating the detached cells from microcarriers in suspension using an efficient, automated methodology, and
c. concentrating large volumes of bulk cell solutions in a timely manner (<5 hours) to maintain cell viability and functionality.
I would like to specifically address a couple of these challenges below:
The harvest unit operation challenge mentioned above is due to the use of proteolytic enzymes (i.e. trypsin) and agitation (i.e. shear forces) during cell dissociation, to dislodge cells from microcarriers, and is currently the industry standard. This unit operation must be done quickly and efficiently, as over-trypsinization and long-term exposure to high shear forces can be harmful to the cells. To address some of the limitations of conventional harvest enzymes, dissociation reagents like TrypLE Select (Gibco-ThermoFisher), have been developed with animal component-free materials, at cGMP grade and in a ready-to-use solution, which will help to ease the future regulatory burden. After the dissociation enzymes have performed their function of detaching the cells from microcarriers, quenching the activity of the enzymes is critical for maintaining the viability of the bulk cell solution for further downstream processing. The industry standard has been to use a solution of 2% fetal bovine serum in phosphate-buffered saline, but the presence of animal-derived components and variation in efficacy of this quench solution pose challenges to its use in robust, reproducible, cGMP-aligned manufacturing processes. Thus, there is a definitive need to develop a xeno-free alternative with validated inactivation of dissociation enzymes to ensure optimal bioprocessing conditions.
Harvesting of cells from microcarriers is then immediately followed by filtration of the cell/microcarrier slurry through a porous mesh that traps the microcarriers while letting cells pass through. Dr. Russotti stressed that confirming complete removal of the microcarriers from the cell therapy product is absolutely critical to ensuring safety for the patient. At the small (0.1L to 5L) to medium (5L to 200L) scale bioreactor size, conventional flow filtration technology has worked, but when the scale increases to 200L and more, the volume of product to process can overwhelm most systems. Upscaled technologies, like continuous flow centrifugation (kSep), have been tested for cell suspensions but will require development and validation for use with cell/microcarrier suspensions. New and promising technology innovation is the development of completely dissolvable microcarriers, which has the potential to obviate the cell/microcarrier separation unit operation in the future, markedly streamlining and simplifying the downstream processing of hMSCs expanded in microcarrier-based bioreactor systems.
While there are several additional manufacturing challenges that will need to be addressed prior to the widespread commercial adoption of hMSC therapies, each of these challenges creates opportunities to develop incrementally and radically innovative products and processes as solutions. NIIMBL represents the perfect platform to approach the challenges in upscaled cell therapy manufacturing through the funding and support of proposals aimed at addressing these industry needs. Currently, there are over 700 clinical trials (clinicaltrials.gov) using hMSCs, a number which has more than doubled since 2014, and hMSCs are poised to be the transistor and microchip equivalent of tomorrow’s Regenerative Medicine technology products. These therapeutic cells are critical to many Regenerative Medicine applications, including cell and gene therapy, bioprinting, and tissue regeneration. The many manufacturing science challenges like those outlined above create the exact motivation for technology innovations with the development of which NIIMBL is tasked.
Given the large patient populations for various target indications (stroke, Graft Versus Host Disease, traumatic brain injury, cardiovascular disease) and dose sizes of 10s to 100s of millions of cells per patient, upscaled manufacturing science technologies will play a critical role in addressing the growing demand for significant volumes of pharmaceutical quality hMSCs. Advancements in Regenerative Medicine are fueling the need for rapid manufacturing science progress, especially with accelerated approval pathways for RegenMed drugs being established around the world. RoosterBio, with our disruptive approach to hMSC manufacturing and use, is uniquely poised to work alongside NIIMBL and other member organizations to push forward hMSC-based therapeutic platforms. We are excited to do our part in revolutionizing Biopharmaceutical manufacturing in the coming years.
