Scalable Manufacturing Solution Needed
Historically, pre-clinical studies and clinical trials have predominantly used hMSCs derived from bone marrow and adipose tissue sources. However, over the last 10 to 15 years, the number of publications and clinical trials in the regenerative medicine field using hMSCs derived from human umbilical cord (hUC-MSCs) as a raw material has significantly increased (1,2,3).
The commercial hUC-MSC therapies that will follow will require hundreds of millions to billions of cells per lot for effective patient dosing (4,5). Traditional 2-dimensional (2D) flask expansion platforms are not cost-effective for such large-scale expansion of cell-based therapeutics. However, 3-dimensional (3D), microcarrier-based, bioreactor systems offer a scalable manufacturing platform that can achieve the lot sizes needed. Here, we demonstrate that hUC-MSCs achieve high cell densities in a 3D bioreactor culture system and maintain their critical quality attributes (CQAs) after harvest.
hUC-MSC Expansion in Scalable Bioreactor Technologies
hUC-MSCs derived from three different donors were cultured in a xeno-free (XF), fed-batch suspension 100mL bioreactor system according to RoosterBio’s bioreactor process recommendations. hUC-MSCs from all three donors reached high cell densities of between 0.8 x 106 and 1.2 x 106 cells/mL after five days of culture (Figure 1). Previous experience with bone marrow-derived hMSCs suggests that bioreactor systems can be scaled to larger volumes such as 3, 15, and 50L systems with similar or even improved cell growth (6).

Figure 1: hUC-MSCs reach new expansion potential.
(A) hUC-MSCs were cultured for five days in a fed-batch bioreactor system with a RoosterReplenish-MSC-XF feed on Day 3. hUC-MSCs reached cell densities of at least 0.8 x 106cells/mL by Day 5. (B) By Day 5, cell-microcarrier aggregation and sampled cell counts indicated that cultures were ready for harvest.
hUC-MSCs Robust Functional Capability
To ensure these cells maintained hMSC CQAs, hUC-MSCs expanded in 3D bioreactors were compared to hUC-MSCs expanded in 2D flasks in a panel of assays (7). After expansion in control flasks (2D) or bioreactors (3D), harvested cells were analyzed for: expansion over a subsequent passage, surface marker expression by flow cytometry, immunomodulatory properties, angiogenic cytokine secretion, and trilineage differentiation (Figure 2). While there was donor-to donor variability, hUC-MSCs that were expanded in 3D bioreactors performed comparably to hUC-MSCs that were expanded in 2D flasks.
This study demonstrates that hUC-MSCs can be expanded in a 3D bioreactor system, reach high cell densities, and maintain their CQAs after harvest. Therefore, hUC-MSCs paired with a bioreactor platform is a system that can be scaled to yield the cell numbers required for product development and commercial therapeutics.
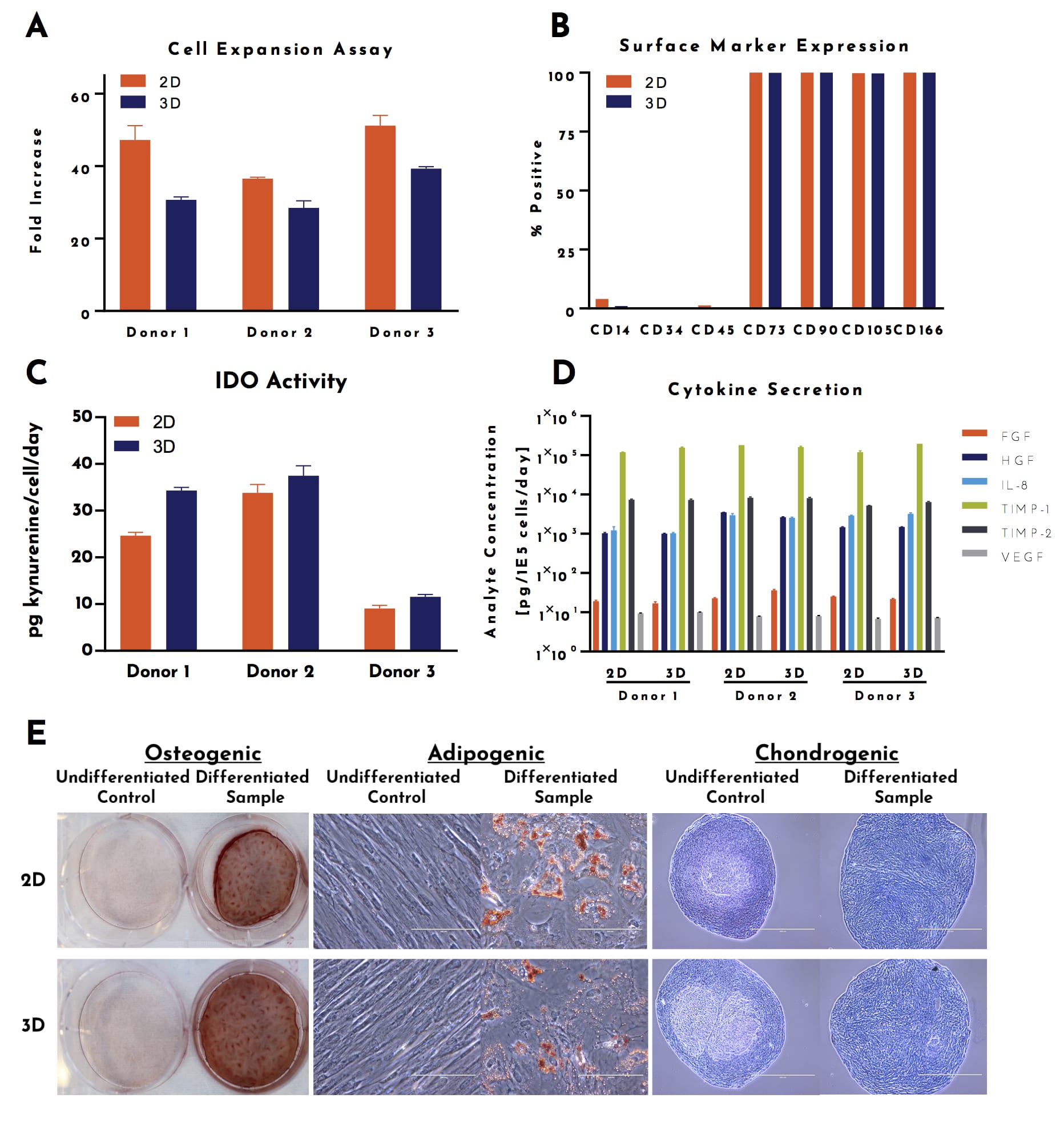
Figure 2: hUC-MSCs maintain critical quality attributes after 3D culture. Freshly harvested cells were plated into functional assays to determine: (A) expansion capacity over a subsequent passage (B) surface marker expression by flow cytometry (representative donor shown) (C) immunomodulatory properties through the secretion of indoleamine-2,3-dioxygenase (D) angiogenic cytokine secretion and (E) trilineage differentiation through media induction (representative donor shown).
References
- Davies JE, Walker JT, Keating A. (2017) Concise Review: Wharton’s Jelly: The Rich, but Enigmatic, Source of Mesenchymal Stromal Cells. Stem Cells Transl Med.,6(7):1620-1630. PubMed
- Zhao J, Yu G, Cai M, Lei X, Yang Y, Wang Q, Zhai X (2018) Bibliometric analysis of global scientific activity on umbilical cord mesenchymal stem cells: a swiftly expanding and shifting focus. Stem Cell Research & Therapy,9(32). PubMed
- Farrance I (2019) Meeting the growing needs of the perinatal RegenMed Industry: the only Umbilical Cord hMSC (hUC-MSC) system designed for today’s translationally focused research and product development. RoosterBio Blog, 18 September 2019. RoosterBlog Post
- Lembong J, Rowley J (2018) Building Effective Multi-Year Process Development Programs: Evolution of Technology Platform Decisions Based on Lot Size. RoosterBio Blog, 15 December 2018. RoosterBlog Post
- Olsen TR, Ng KS, Lock LT, Ahsan T, Rowley JA (2018) Peak MSC – are we there yet? Front. Med.,21 June 2018. https://www.ncbi.nlm.nih.gov/pubmed/29977893
- Kirian RD, Wang D, Takacs J, Tsai A, Cruz K, Rosello F, Cox K, Hashimura Y, Lembong J, Rowley JA, Jung S (2019) Scaling A Xeno-Free Fed-Batch Microcarrier Suspension Bioreactor System From Development to Production Scale for Manufacturing XF hMSCs. Cytotherapy, 2019 May 1;21(5):S71-2. RoosterPoster Link
- Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop D, & Horwitz E (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy,8(4):315-317. http://www.ncbi.nlm.nih.gov/pubmed/16923606.
