
When I was a beginning graduate student, I was transfecting human mesenchymal stem/stromal cells (hMSCs) with polyethyleneimine (PEI), a common polymer used in genetic engineering procedures. I wondered what effect this large polymer had on cell phenotype and differentiation potential once it was endocytosed. I found that transfection with PEI:GFP-pDNA polyplexes changed both a marker for hMSC’s stemness and their differentiation potential. [1]
Genetic engineering techniques that enable high loading efficiencies while maintaining MSC’s key critical quality attributes is still a main goal for many advanced therapy product developers.
To address this barrier, RoosterBio and Portal Biotechnologies have collaborated to develop a turn-key “Boosting” protocol to load MSCs with exogenous cargoes.
The protocol combines RoosterVial™–hMSCs, RoosterNourish™-MSC-XF, and RoosterGEM™ with Portal’s Gateway™ system for high-efficiency loading of cargoes like mRNA, pDNA, peptides, proteins, antibodies, small molecules, and ribonucleoprotein particles (RNPs). Portal developed the Gateway™ system to “Boost” cells via mechanical delivery of diverse molecules, an approach that has been shown to be more “cell friendly” than other loading approaches (Figure 1). [2]
Figure 1 (below). Schematic representation of Portal’s “Boosting” technology which uses pores in specialized silicon membranes to temporarily mechanically permeabilize cells allowing cargo to enter their cytoplasm.

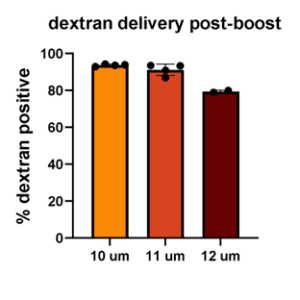
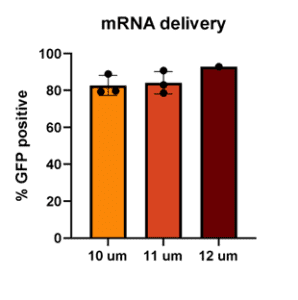
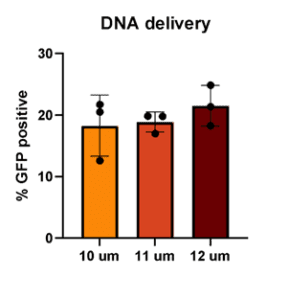
To develop this protocol, a series of collaborative optimization experiments were performed, resulting in compelling data demonstrating that hMSCs could be “Boosted” to contain fluorescently labeled-dextran as an example macromolecule, GFP-mRNA, and GFP-pDNA. Greater than 80% of hMSCs contained fluorescently labeled-dextran post boosting and expressed GFP from mRNA, while maintaining greater than 75% viability. Intriguingly, approximately 20% of hMSCs expressed GFP from pDNA while also maintaining high viability. This result was unexpected, as it is known that “Boosting” typically delivers cargo into the cytoplasm, and pDNA alone typically does not efficiently translocate to the nucleus for expression. Interestingly, “boosting” with 3 different pore sizes produced similar transfection efficiencies. This contrasts with other primary cell types that Portal has tested, which typically require one specific pore size for maximum efficiency. In general, RoosterGEM™ increased loading efficiency without decreasing viability compared to other reagents.
Figure 2 (below). Percent delivery of dextran (top left), mRNA (top right), and pDNA (bottom left) to hMSC with 10µm, 11µm, or 12µm pore size membranes in the Gateway System.
RoosterVial-hMSCs, RoosterNourish-MSC-XF, and RoosterGEM are available in research use only (RUO) and current good manufacturing process (cGMP) grades. Portal’s Gateway device is built for early-stage product and process development, and they have developed a module-based approach where their “Boosting” technology can readily be incorporated into the bioreactor workflows of many leading bioprocessing companies. Together, RoosterBio’s and Portal’s technologies can provide a clinically relevant and scalable approach to manufacturing your engineered-cell product.
This protocol builds on the continuing list of applications for RoosterGEM including pDNA transfection, [3, 4] lentiviral transduction, [5] and AAV transduction of MSCs. [6] Other applications include the use of RoosterGem to enhance primary immune cell transduction, [7] and adenovirus transfection efficiencies. [8]
How could you use RoosterBio’s scalable portfolio of MSC and exosome manufacturing products with Portal’s “Boost” technology to improve your product development? Could you benefit from high cargo loading efficiencies and lower cost of goods?
I personally find the combinations of these technologies to be an elegant solution to loading hMSCs with diverse cargo. My hope is that you can leverage the protocol from this exciting collaboration to help accelerate your own hMSC and exosome research, product, and process development.
References
- King, W. J.; Kouris, N. A.; Choi, S.; Ogle, B. M.; Murphy, W. L., Environmental parameters influence non-viral transfection of human mesenchymal stem cells for tissue engineering applications. Cell Tissue Res 2012, 347 (3), 689-99. 10.1007/s00441-011-1297-0
- Sharei, A.; Zoldan, J.; Adamo, A.; Sim, W. Y.; Cho, N.; Jackson, E.; Mao, S.; Schneider, S.; Han, M. J.; Lytton-Jean, A.; Basto, P. A.; Jhunjhunwala, S.; Lee, J.; Heller, D. A.; Kang, J. W.; Hartoularos, G. C.; Kim, K. S.; Anderson, D. G.; Langer, R.; Jensen, K. F., A vector-free microfluidic platform for intracellular delivery. Proc Natl Acad Sci U S A 2013, 110 (6), 2082-7. 10.1073/pnas.1218705110
- Doolin, M. T.; Willstaedt, T. M.; Rowley, J. A., Mesenchymal Stem/Stromal Cells: OPTIMIZATION OF TRANSIENT TRANSFECTION OF MESENCHYMAL STROMAL CELLS WITH PLASMID DNA OR MESSENGER RNA. Cytotherapy 2022, 24 (5), S59. 10.1016/S1465-3249(22)00196-7
- King, B. How to Achieve High mRNA Transfection Efficiencies in Primary Adherent Cells. https://www.roosterbio.com/blog/how-to-achieve-high-mrna-transfection-efficiencies-in-primary-adherent-cells/.
- Willstaedt, T. M.; Walde, A.; Brennan, J.; Rowley, J. A.; Adlerz, K., Development of an optimized lentiviral transduction medium and process to manufacture genetically modified MSC working cell banks. Cytotherapy 2021, 23 (5), S45. 10.1016/S1465324921003169
- Doolin, M., Willstaedt, T., Boychyn, M., Rowley, J. AAV Transduction of MSCs is Enhanced by Genetic Engineering Medium. https://www.roosterbio.com/resource/aav-transduction-of-mscs-is-enhanced-by-genetic-engineering-medium/.
- Doolin, M., Carson, J. RoosterGEM™ Shines on Lentiviral Transduction of T Cells. https://www.roosterbio.com/blog/roostergem-shines-on-lentiviral-transduction-of-t-cells/.
- RoosterBio The Role hMSCs Play in Bioengineering Solutions for Military Medicine. https://tinyurl.com/4d7weuvf.
