Designed for product developers and translational researchers needing quick access to billions, not millions, of hMSCs.
Recommended Products
Overview
RoosterVial™-hUC-XF are Human Umbilical Cord Derived Mesenchymal Stem/Stromal Cells (hUC-MSCs) isolated from the perivascular Wharton’s Jelly region of the human umbilical cord and expanded to a population doubling level (PDL) of 14-20. Available in both 1M or high-volume 10M cell per vial formats to meet your needs throughout product development.
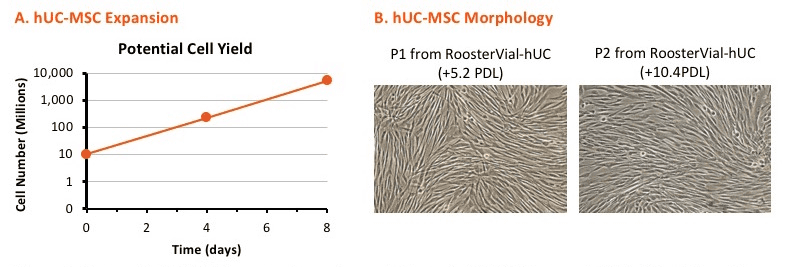
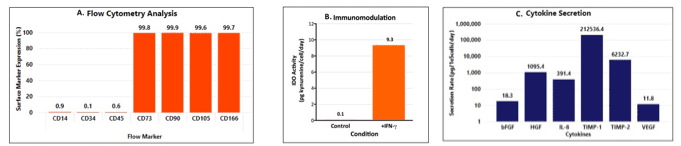
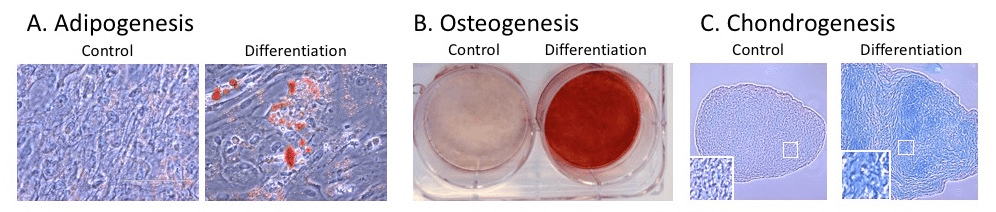
Each lot is release tested to expand >10-fold within 4 to 6 days in a media exchange free process when paired with RoosterNourish™-MSC-XF bioprocess medium and conforms to ISCT minimal criteria for hMSC identity including cell surface marker expression and tri-lineage differentiation potential, expansion potential, and sterility. Accompanying QC brief contains industry-leading functional characterization including immunomodulatory potential (IDO assay) and angiogenic cytokine secretion, giving added confidence in quality, reproducibility, and ease of integration.
Designed for robust, reproducible, standardized XF scale-up and supported by our media exchange free 2D and 3D bioreactor processes and RoosterNourish™-MSC-XF expansion media, XF RoosterVial™-hUC can efficiently and reproducibly get you the millions or billions of hMSCs to accelerate your Research and Product Development.
*RoosterVial-hUC-XF manufactured and sold by RoosterBio, INC and supported by licensed technology from Tissue Regeneration Therapeutics Inc. (TRT) core technology and patent family: US 8,790,923; US 8,278,102; US 7,547,546; US 9,611,456; US 9,611,456; US 8,481,311; US 9,611,456.
Product Features
- Available in GMP Grade for Use in Clinical Manufacturing
- 1M (C43001UC) or 10M (C43002UC) cryopreserved, xeno-free hUC-MSCs with multiple available donors
- Supplied, easy-to-follow expansion protocols, saving you months of process development time.
- Supported by 1st in class hMSC identity and functional characterization
- > 10-fold, media exchange free expansion in 4 to 5 days when cultured with RoosterNourish™-MSC-XF
- Manufactured with cGMP compatible processes
- Try the complete hMSC and media system with RoosterKit™-hUC-1M-XF or XF hUC-hMSC Donor Screening Kit.
Intended Use: Research Use Only
Protocols and Information
- C43001UC, C43002UC, C45010UC Safety Data Sheet
- RoosterVial™-hUC-1M-XF (C43001UC) Expansion Protocol
- RoosterVial™-hUC-10M-XF (C43002UC) Expansion Protocol
Login to search Certificates of Analysis and Quality Control Briefs.